Revolutionising Tuberculosis Treatment test
Innovative drug delivery approaches
Ashok Aspatwar, Adjunct Professor, Tampere University
Rarchita Sharma, Researcher, SVKM's Institute of Pharmacy
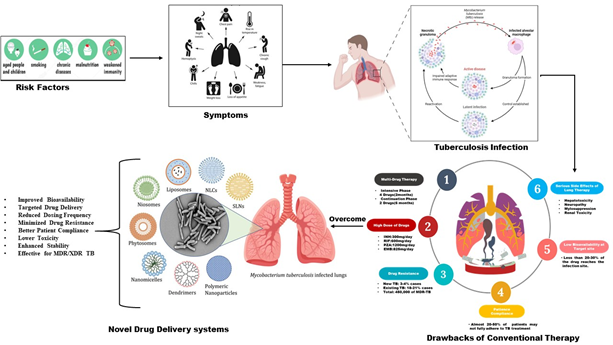
Tuberculosis (TB) treatment faces challenges such as poor drug bioavailability, prolonged therapy, and patient non-compliance. Innovative drug delivery systems, including inhalable nanoparticles, lipid-based carriers, and microneedle patches, are transforming TB management. These technologies enhance targeted drug delivery, reduce systemic toxicity, and improve patient adherence by enabling controlled and localised release of anti-TB drugs.
Introduction
Tuberculosis (TB) is a contagious and often deadly infectious disease caused by the bacterium Mycobacterium tuberculosis. While it primarily affects the lungs (pulmonary TB), the infection can also spread to other parts of the body, such as the lymph nodes, bones, kidneys, and brain (extrapulmonary TB). TB remains one of the top 10 causes of death worldwide, despite being both preventable and curable. According to the World Health Organization (WHO), approximately 10 million people fall ill with TB each year, and over 1.3 million die from the disease. TB transmission occurs through the air when an infected person coughs, sneezes, or speaks, making it a public health concern, especially in overcrowded and under-resourced areas. The burden of TB is disproportionately heavy in low- and middle-income countries, where access to healthcare is limited, and the disease is often exacerbated by conditions such as HIV, malnutrition, and poverty.
The standard treatment for TB has been in place for decades and typically involves a combination of antibiotics, including isoniazid, rifampicin, pyrazinamide, and ethambutol, taken daily over a course of at least six months. For patients with multidrug-resistant TB (MDR-TB) or extensively drug-resistant TB (XDR-TB), the treatment duration can extend to 18 to 24 months and requires the use of second-line drugs that are more toxic, more expensive, and often less effective. The prolonged duration of therapy often leads to poor patient adherence, particularly in regions with inadequate healthcare infrastructure. Patients often struggle with taking multiple medications daily, coping with adverse drug reactions such as liver toxicity, gastrointestinal problems, and peripheral neuropathy, and maintaining motivation over such a prolonged period. These factors contribute significantly to incomplete treatment, leading to treatment failure and the emergence of drug-resistant TB strains, which are harder to treat and control.
To address these significant drawbacks, the focus of TB management has increasingly shifted towards the development of novel drug delivery systems. These advanced delivery methods aim to enhance efficacy, reduce toxicity, and improve the accessibility of TB treatment. Instead of relying solely on oral pills or injections, researchers are exploring innovative technologies that control how, when, and where anti-TB drugs are released in the body. These include nanoparticle-based delivery systems, inhalable formulations, long-acting implants, and transdermal microneedle patches. Each of these approaches is designed to overcome specific limitations of conventional therapy and to offer a more patient-centered treatment strategy.
The benefits of these novel drug delivery systems over conventional therapy are substantial. They offer controlled, sustained, or targeted drug release, which not only improves the pharmacokinetics and bioavailability of the medication but also reduces the frequency of administration. This, in turn, leads to better patient adherence, fewer missed doses, and a lower risk of developing drug resistance. Additionally, these systems often reduce the side effects by minimising systemic exposure and targeting the drug at the site of infection. Many of these delivery methods are also more user-friendly and adaptable to community-based or self-administered models, making them particularly suitable for resource-limited settings where access to consistent healthcare is a challenge. However, these novel drug delivery technologies have the potential to transform the current TB treatment paradigm, making it shorter, safer, and more effective for patients across diverse populations.
Pathogenesis of Tuberculosis
Tuberculosis infection progresses through five key stages. Initially, inhaled bacilli reach the lung alveoli, where alveolar macrophages often neutralise them within 3–8 weeks. If this defense fails, the bacteria multiply inside immature macrophages, leading to the absorption stage. As the infection progresses, an immune response triggers inflammation, tissue damage (caseous necrosis), and the formation of tubercles. While bacterial growth stops during this latent phase, some bacilli may survive or even continue replicating. In progressive cases, liquefaction of the tubercle core leads to the formation of a cavity, enabling rapid bacterial multiplication and spread within the lungs. Finally, in the extrapulmonary stage, the bacteria disseminate via the lymphatic or blood systems, potentially infecting organs such as the kidneys, brain, bones, and gastrointestinal tract.
Novel Drug Delivery Systems
Tuberculosis (TB) treatment traditionally involves long-term, multi-drug regimens that can lead to poor patient compliance and drug resistance. To overcome these challenges, novel drug delivery systems (NDDS) are being explored to enhance therapeutic efficacy, reduce dosing frequency, and minimise side effects. Nanotechnology-based carriers, such as liposomes, nanoparticles, solid lipid nanoparticles, dendrimers, and microspheres, offer controlled and targeted delivery of anti-TB drugs, thereby improving their bioavailability and penetration into infected macrophages and granulomas. Inhalable formulations deliver drugs directly to the lungs, enhancing local concentration and reducing systemic toxicity. Stimuli-responsive systems activated by pH, temperature, or enzymes allow for site-specific drug release, which is particularly effective against the dormant bacilli residing in hypoxic, acidic tubercles. Furthermore, oral, injectable, and transdermal systems are being modified with advanced polymers for sustained drug release. These innovations represent a promising leap in TB management, aiming for shorter treatment durations, better patient adherence, and reduced drug resistance.
1. Liposomes
Liposomes are spherical vesicles with a lipid bilayer that can encapsulate both hydrophilic and lipophilic drugs. In TB treatment, liposomes enhance drug delivery by targeting infected macrophages, where Mycobacterium tuberculosis typically resides. Their biocompatibility and ability to protect drugs from degradation make them ideal carriers. Liposomal formulations can also reduce systemic side effects and allow sustained drug release, thereby improving patient compliance and treatment effectiveness.
2. Nanoparticles
Nanoparticles, made from polymers, metals, or lipids, are tiny carriers (1–1000 nm) that can improve the delivery of anti-TB drugs. They penetrate deeply into tissues and cells, including macrophages and granulomas, enhancing drug accumulation at infection sites. Polymer-based nanoparticles, such as those made from poly (lactic-co-glycolic acid) (PLGA), can provide sustained release, improve drug solubility, and protect drugs from early metabolism. They are particularly useful in targeting latent TB bacilli.
3. Solid Lipid Nanoparticles (SLNs)
SLNs are composed of solid lipids and provide a stable, biocompatible platform for drug delivery. They combine the advantages of liposomes and polymeric nanoparticles, offering controlled release, high drug loading, and long-term stability. SLNs can enhance the oral bioavailability of anti-TB drugs and improve their transport across biological membranes. Their ability to localise in macrophages makes them promising carriers for both active and latent TB treatment.
4. Dendrimers
Dendrimers are highly branched, tree-like synthetic polymers with a high surface area for drug attachment. Their structure allows for precise control over size, shape, and surface functionality. In TB therapy, dendrimers can be engineered to target macrophages, enhance drug solubility, and allow for controlled release. Their multivalency enables them to carry multiple drug molecules or targeting ligands, improving the specificity and efficacy of the treatment.
5. Microspheres
Microspheres are spherical particles, typically in the micrometer range, made from biodegradable polymers such as PLGA. They can encapsulate anti-TB drugs and provide controlled, long-term drug release over days or weeks. This sustained release reduces the frequency of dosing and improves patient adherence. Microspheres are especially useful in pulmonary drug delivery systems, where they can be inhaled to deliver drugs directly to the lungs.
6. Inhalable Formulations
Inhalable drug delivery systems allow direct administration of anti-TB drugs to the lungs, the primary site of infection. This approach increases drug concentration at the infection site, reduces systemic exposure, and improves efficacy. Inhalable powders or aerosols can be formulated using nanoparticles or microspheres, ensuring deep lung penetration and prolonged retention. This method is particularly beneficial in targeting drug-resistant or dormant TB bacilli within granulomas.
7. Stimuli-Responsive Systems
These advanced systems release drugs in response to specific internal stimuli such as pH, enzymes, or temperature. For TB, stimuli-responsive carriers can exploit the acidic, hypoxic environment of granulomas to trigger drug release specifically at infected sites. This targeted release minimises damage to healthy tissues and enhances drug concentration where it is most needed, making treatment more effective against persistent and latent infections.
8. Modified Oral, Injectable, and Transdermal Systems
Traditional drug delivery routes like oral, injectable, and transdermal are being enhanced with novel polymers and nanotechnology. Modified oral tablets may use enteric coatings or mucoadhesive systems for controlled release. Injectable nanoformulations can provide long-acting drug release over weeks or months. Transdermal patches with micro-needles or nanoparticles enable non-invasive, sustained drug delivery. These systems help in maintaining steady drug levels and reducing treatment duration and pill burden.
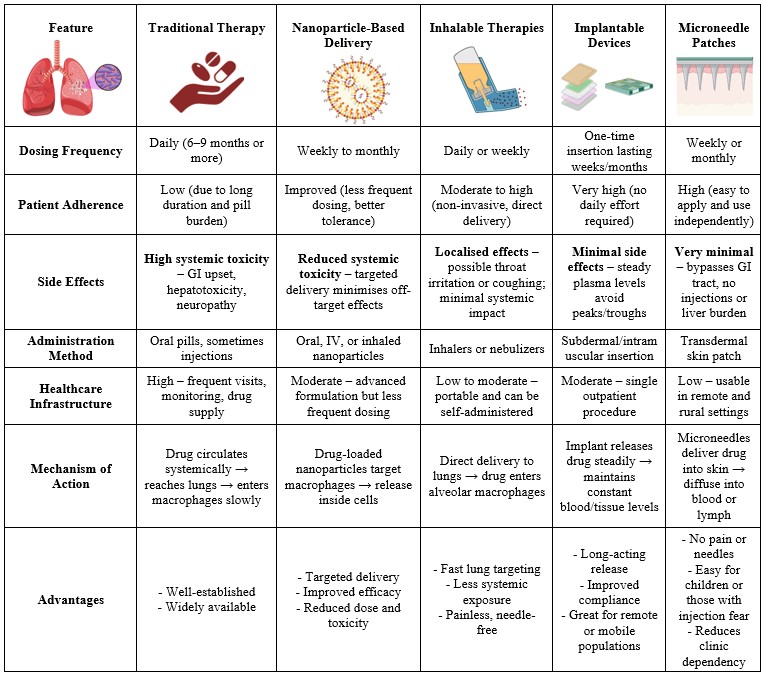
Conclusion:
Novel drug delivery systems offer promising advancements in tuberculosis treatment by enhancing drug targeting, improving bioavailability, reducing side effects, and enabling sustained release. These innovative approaches can shorten treatment duration, improve patient adherence, and help combat drug-resistant TB, marking a significant step toward more effective disease management.